81. Direct Electrooxidation of Ethylene to Ethylene Glycol over 90% Faradaic Efficiency Enabled by Cl– Modification of the Pd Surface
An-Zhen Li, Xiongbo Wang, Shuwei Li, Bo-Jun Yuan, Xi Wang, Ruo-Pu Li, Liang Zhang, Bi-Jie Li*, Haohong Duan*
J. Am. Chem. Soc. 2025,147, 12, 10493–10503
DOI:10.1021/jacs.4c18345
Abstract
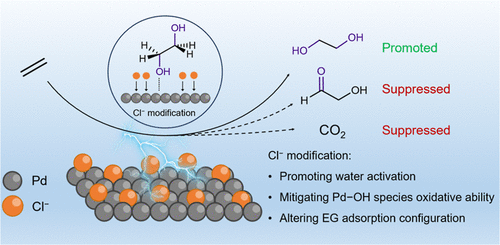
Direct electrochemical ethylene-to-ethylene glycol (C2H4-to-EG) conversion can potentially reduce the consumption of fossil fuels and the emission of carbon dioxide (CO2) compared with the traditional thermo-catalytic approach. Palladium (Pd) prepared by electrodeposition is represented as a promising electrocatalyst; however, it exhibits low Ethylene glycol (EG) current density (<4 mA cm–2), Faradaic efficiency (<60%), and productivity (<10 μmol h–1), hindering practical applications. Herein, we report a nanodendrite palladium catalyst supported on a large-area gas diffusion electrode. This catalyst gives high EG current density (12 mA cm–2) and productivity (227 μmol h–1) but low Faradaic efficiency (65%). With further Cl– ions modification, Faradaic efficiency increased to a record-high value of 92%, and EG current density (18 mA cm–2) and productivity (∼340 μmol h–1) were also promoted. Experimental data suggest that the strong electron-withdrawing feature of Cl– reduces the oxidation ability of in situ generated Pd–OH species, inhibiting EG overoxidation to glycol aldehyde. Meanwhile, Cl– alters EG adsorption configuration─from parallel and dual-site coordination to vertical and single-site coordination─over the Pd surface, thus preventing C–C bond cleavage of EG to CO2. In addition, Cl– adsorption facilitates the generation of Pd–OH active species to improve catalytic activity. This work demonstrates the great potential of surface ion modification for improving activity and selectivity in direct electrochemical C2H4-to-EG conversion, which may have implications for diverse value-added chemicals electrosynthesis.