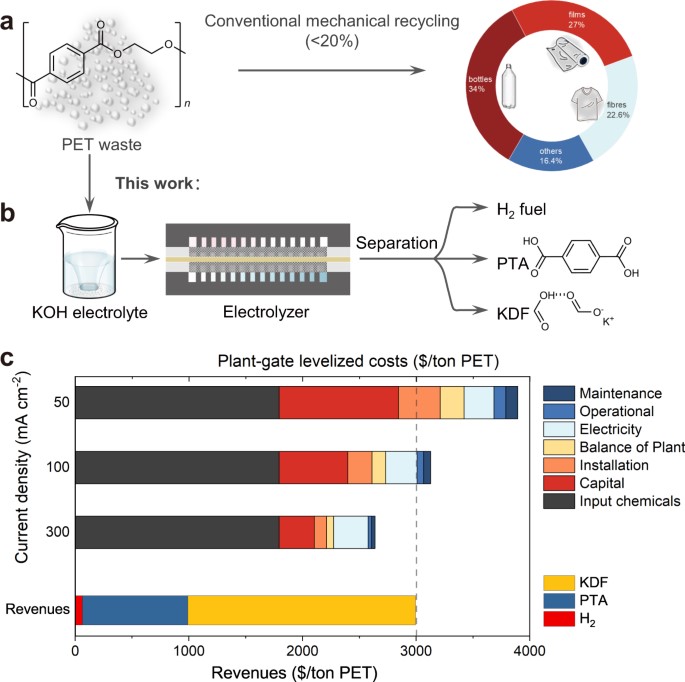
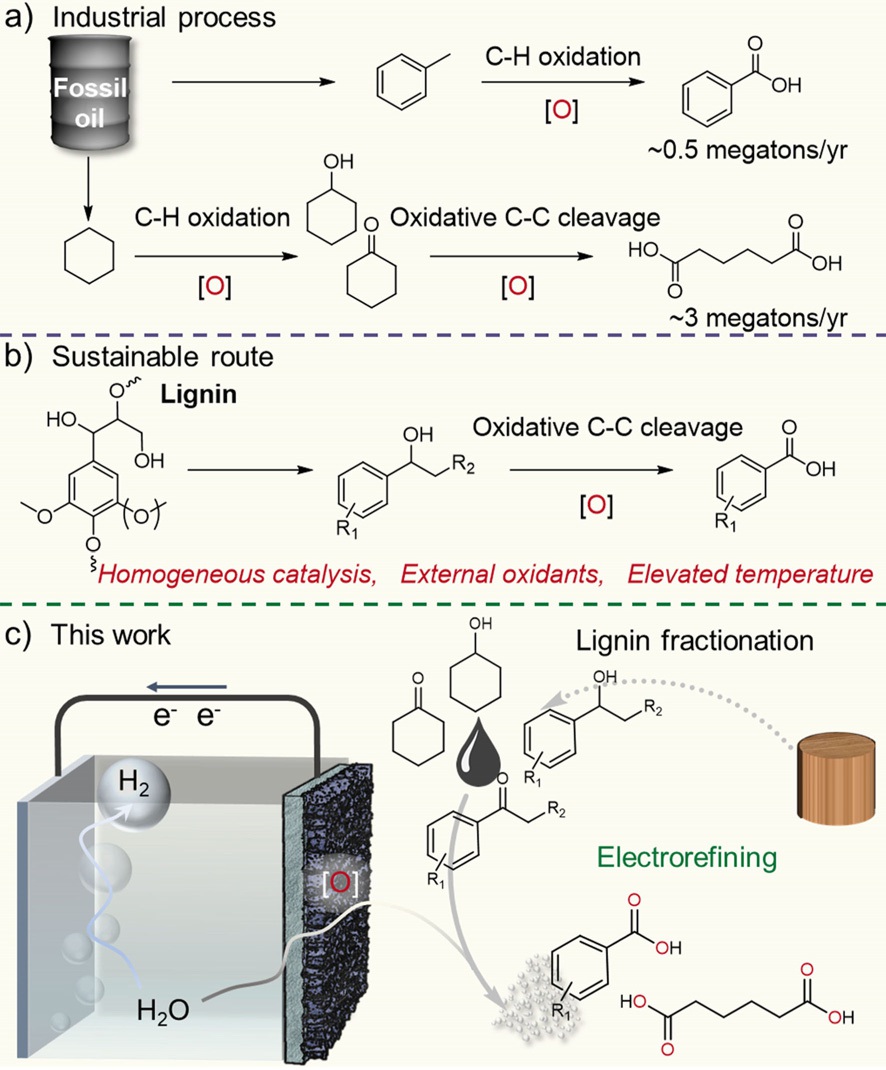
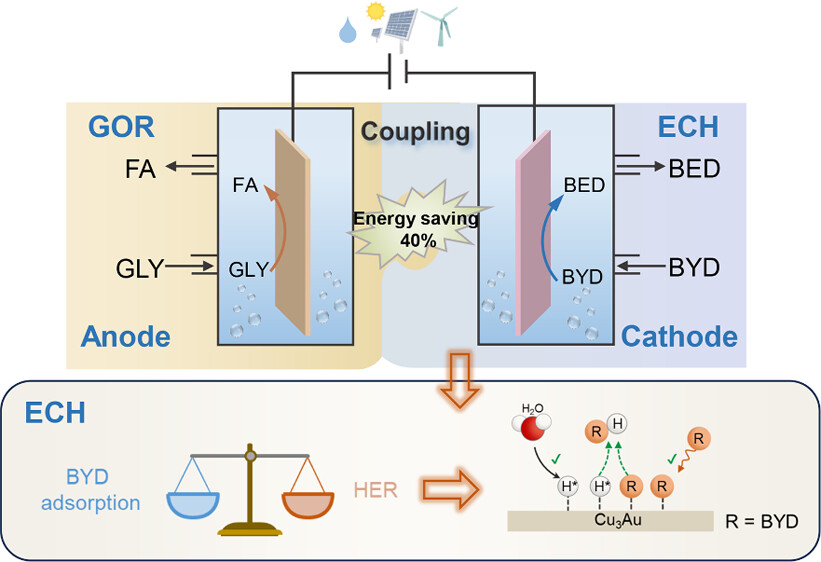
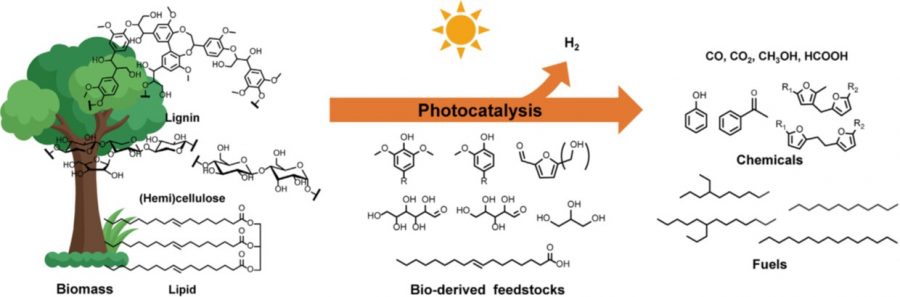
26. Atomically Dispersed Pt-N3C1 Sites Enabling Efficient and Selective Electrocatalytic C-C Bond Cleavage in Lignin Models under Ambient Conditions
Cui, T., Ma, L., Wang, S., Ye, C., Liang, X., Zhang, Z., Meng, G., Zheng, L., Hu, H. S., Zhang, J.*, Duan, H.*, Wang, D.*, Li, Y.
J. Am. Chem. Soc. 2021, 143 (25), 9429-9439.
DOI: 10.1021/jacs.1c02328
Abstract
Selective cleavage of C–C linkages is the key and a challenge for lignin degradation to harvest value-added aromatic compounds. To this end, electrocatalytic oxidation presents a promising technique by virtue of mild reaction conditions and strong sustainability. However, the existing electrocatalysts (traditional bulk metal and metal oxides) for C–C bond oxidative cleavage suffer from poor selectivity and low product yields. We show for the first time that atomically dispersed Pt–N3C1 sites planted on nitrogen-doped carbon nanotubes (Pt1/N-CNTs), constructed via a stepwise polymerization–carbonization–electrostatic adsorption strategy, are highly active and selective toward Cα–Cβ bond cleavage in β-O-4 model compounds under ambient conditions. Pt1/N-CNTs exhibits 99% substrate conversion with 81% yield of benzaldehyde, which is exceptional and unprecedented compared with previously reported electrocatalysts. Moreover, Pt1/N-CNTs using only 0.41 wt % Pt achieved a much higher benzaldehyde yield than those of the state-of-the-art bulk Pt electrode (100 wt % Pt) and commercial Pt/C catalyst (20 wt % Pt). Systematic experimental investigation together with density functional theory (DFT) calculation suggests that the superior performance of Pt1/N-CNTs arises from the atomically dispersed Pt–N3C1 sites facilitating the formation of a key Cβ radical intermediate, further inducing a radical/radical cross-coupling path to break the Cα–Cβ bond. This work opens up opportunities in lignin valorization via a green and sustainable electrochemical route with ultralow noble metal usage.