Congratulations to Kaiyue Ji on the article about Electrocatalytic Hydrogenation of 5-Hydroxymethylfurfural Promoted by a Ru1Cu Single-Atom Alloy Catalyst being accepted by Angewandte Chemie!
Kaiyue Ji,Ming Xu,Si-Min Xu,Ye Wang,Ruixiang Ge,Xiaoyu Hu,Xiaoming Sun,Haohong Duan*
DOI: 10.1002/anie.202209849
Abstract
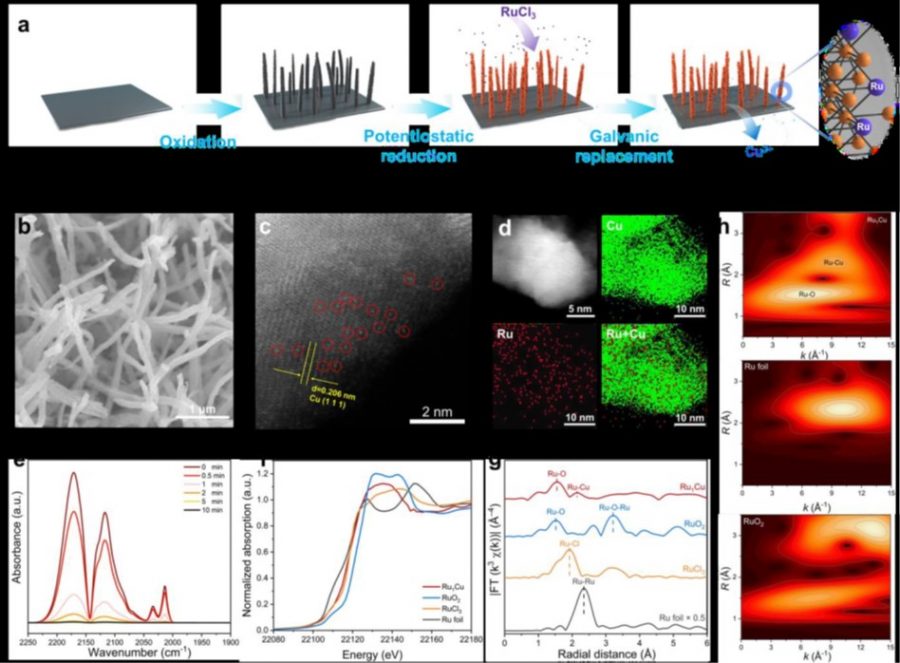
Electrochemical reduction of biomass-derived 5-hydroxymethylfurfural (HMF) represents an elegant route toward sustainable value-added chemicals production that circumvents the use of fossil fuel and hydrogen. However, the reaction efficiency is hampered by the high voltage and low activity of electrodes (Cu, Bi, Pb). Herein, we report a Ru 1 Cu single-atom alloy (SAA) catalyst with isolated Ru atoms on Cu nanowires that exhibits an electrochemical reduction of HMF to 2,5-dihydroxymethylfuran (DHMF) with promoted productivity (0.47 vs. 0.08 mmol·cm -2 ·h -1 ) and faradic efficiency (FE) (85.6 vs. 71.3%) at – 0.3 V (vs. RHE) compared with Cu counterpart. More importantly, the FE (87.5%) is largely retained at high HMF concentration (100 mM). Kinetic studies by using combined electrochemical techniques suggest disparate mechanisms over Ru 1 Cu and Cu, revealing that single-atom Ru promotes the dissociation of water to produce H* species that effectively react with HMF via an electrocatalytic hydrogenation (ECH) mechanism.