59. Unraveling the potential-dependent structure evolution in CuO forelectrocatalytic biomass valorization
Ye Wang, Ming Xu, Xi Wang, Ruixiang Ge, Yu-Quan Zhu, An-Zhen Li, Hua Zhou, Fengen Chen, Lirong Zheng, Haohong Duan*
Sci. Bull. 2023, 68, 23, 2982-2992.
DOI: 10.1016/j.scib.2023.09.033
Abstract
Electrocatalytic oxidation of renewable biomass (such as glucose) into high-value-added chemicals provides an effective approach to achieving carbon neutrality. CuO-derived materials are among the most
promising electrocatalysts for biomass electrooxidation, but the identification of their active sites under
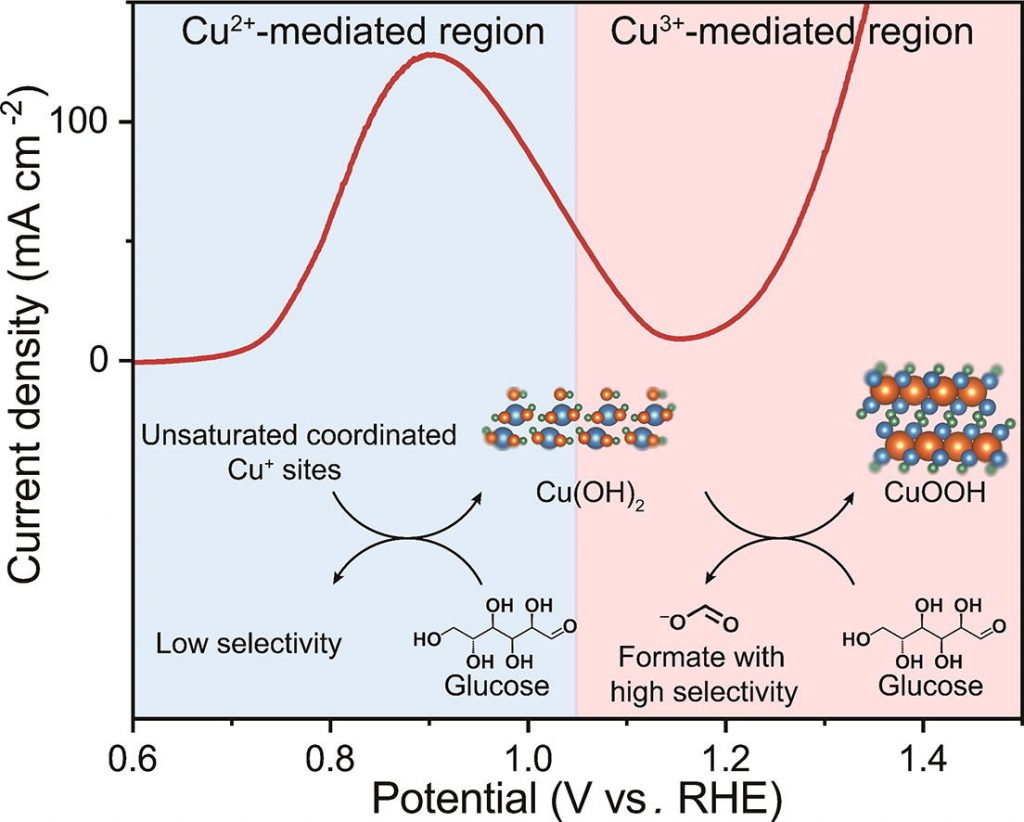
electrochemical conditions remains elusive. Herein, we report a potential-dependent structure evolution
over CuO in the glucose oxidation reaction (GOR). Through systematic electrochemical and spectroscopic
characterizations, we unveil that CuO undergoes Cu2+/Cu+ and Cu3+/Cu2+ redox processes at increased
potentials with successive generation of Cu(OH)2 and CuOOH as the active phases. In addition, these
two structures have distinct activities in the GOR, with Cu(OH)2 being favorable for aldehyde oxidation,
and CuOOH showed faster kinetics in carbon–carbon cleavage and alcohol/aldehyde oxidation. This work
deepens our understanding of the dynamic reconstruction of Cu-based catalysts under electrochemical
conditions and may guide rational material design for biomass valorization.